Description
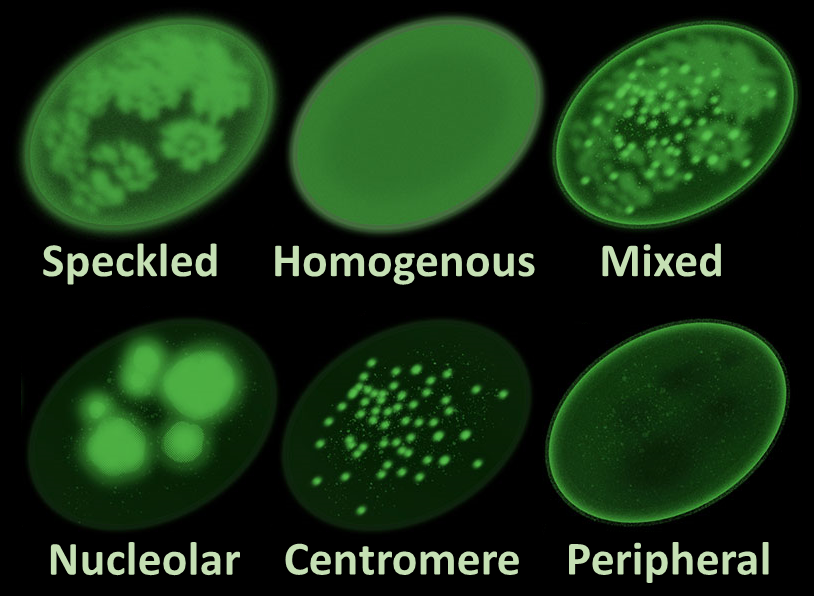
The antinuclear antibody (ANA) test, particularly when performed using indirect immunofluorescence assay (IFA) on HEp-2 cells, is a common diagnostic tool used to detect the presence of antibodies that target various components of the cell nucleus. These antibodies are characteristic of autoimmune diseases such as systemic lupus erythematosus (SLE), Sjögren’s syndrome, and systemic sclerosis.
When the ANA test is conducted using IFA on HEp-2 cells, the result typically includes information about the titer or dilution at which the antibodies were detected. For example, a report might indicate a result such as “positive at 1:320 dilution.”
Here’s what this means:
- Dilution: The ANA test is usually performed by diluting the patient’s serum (the liquid part of the blood) in a series of steps. Each step involves further dilution of the serum with a buffer solution. The dilution is expressed as a ratio, where the first number represents the amount of serum and the second number represents the amount of buffer solution. For example, in a 1:320 dilution, the serum has been diluted 320 times with the buffer solution.
- Titer: The titer refers to the highest dilution at which the antibodies are still detectable. In the example of a result “positive at 1:320 dilution,” it means that the antibodies were still detectable when the serum was diluted up to 1:320. If the antibodies are detected at a lower dilution, such as 1:40 or 1:80, it suggests a higher antibody concentration and may be indicative of a stronger immune response.
The interpretation of ANA results, including the significance of titers, requires consideration of various factors, including the patient’s symptoms, medical history, and presence of other autoantibodies. A positive ANA test result alone does not necessarily indicate the presence of an autoimmune disease but rather suggests the need for further evaluation by a healthcare provider, typically a rheumatologist, to determine the underlying cause of the positive result.


Reviews
There are no reviews yet.